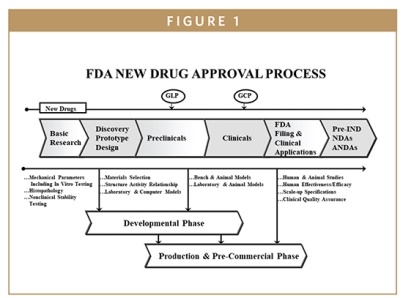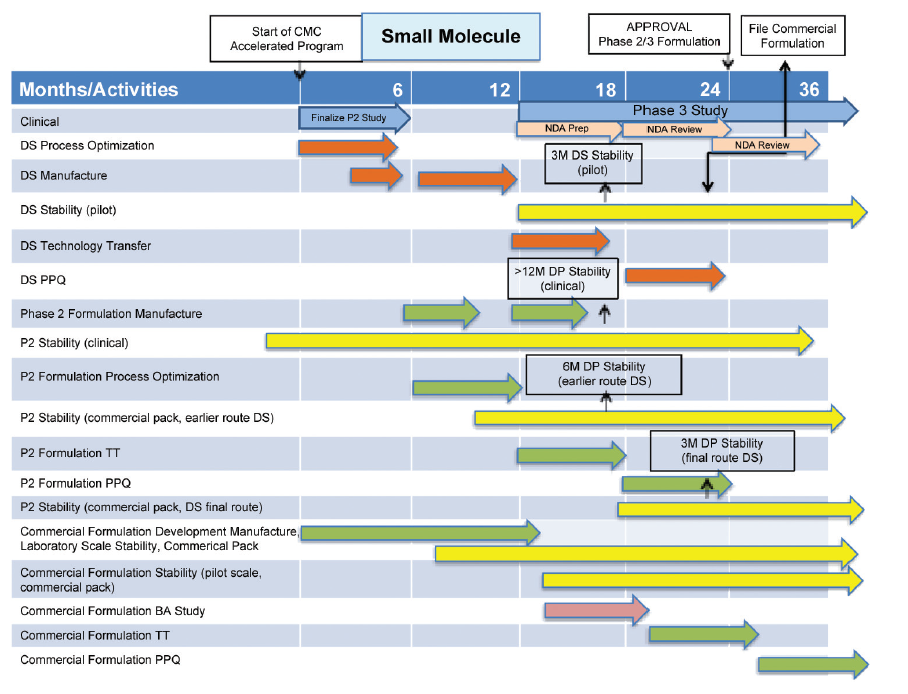
CMC Considerations when a Drug Development Project is Assigned Breakthrough Therapy Status | Pharmaceutical Engineering
EXECUTIVE SUMMARY – Clinical Trials Planning Meeting on Strategies for Integrating Biomarkers into Clinical Development of New
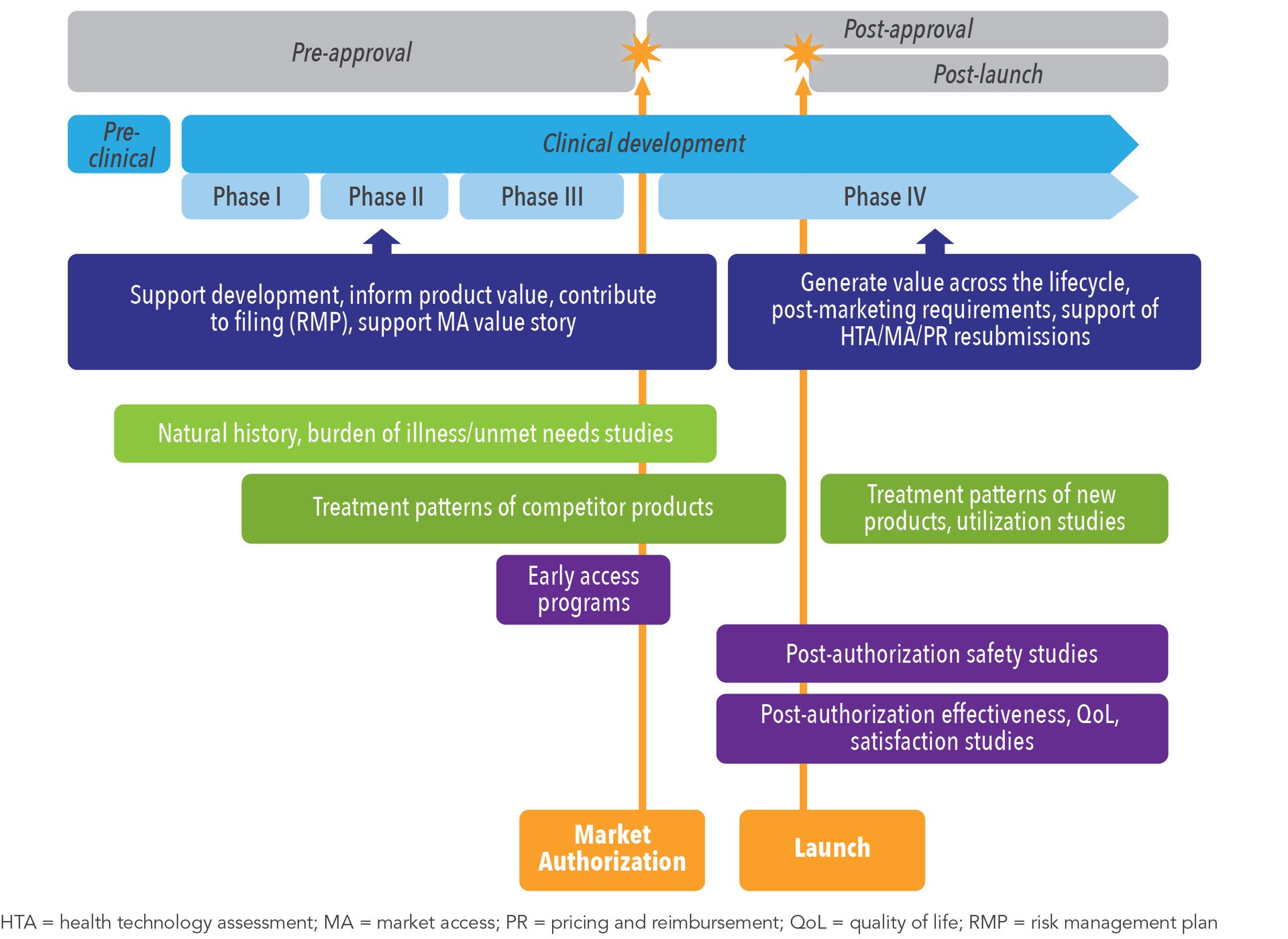
White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera
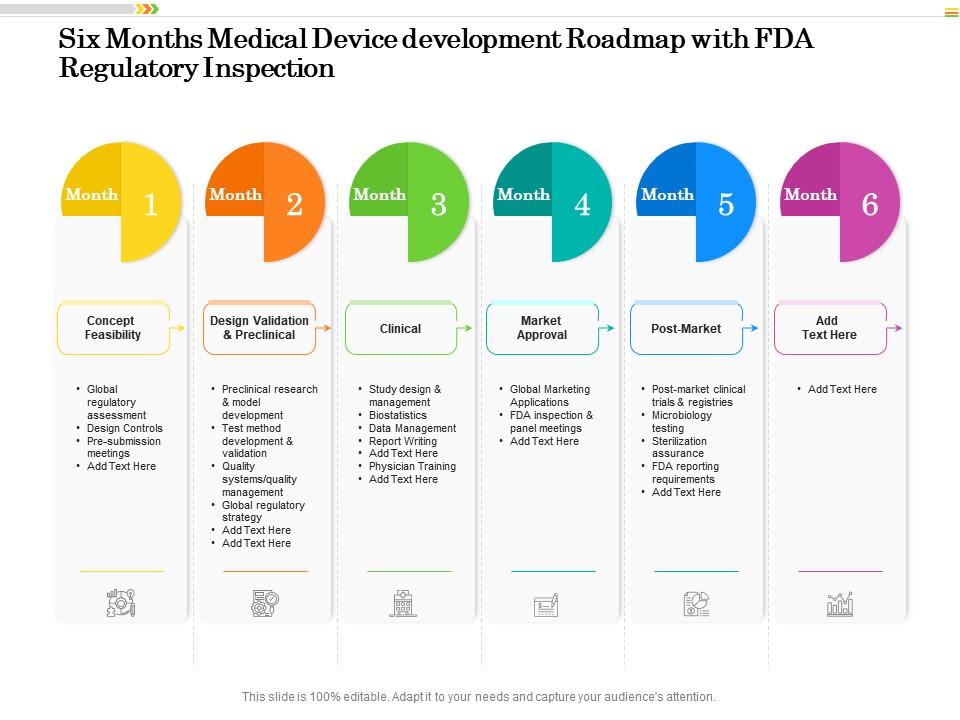
Six Months Medical Device Development Roadmap With FDA Regulatory Inspection | PowerPoint Slides Diagrams | Themes for PPT | Presentations Graphic Ideas

Column - 5 Top Annual Plan Medical Device Design and Development Process Improvements - MedTech Intelligence

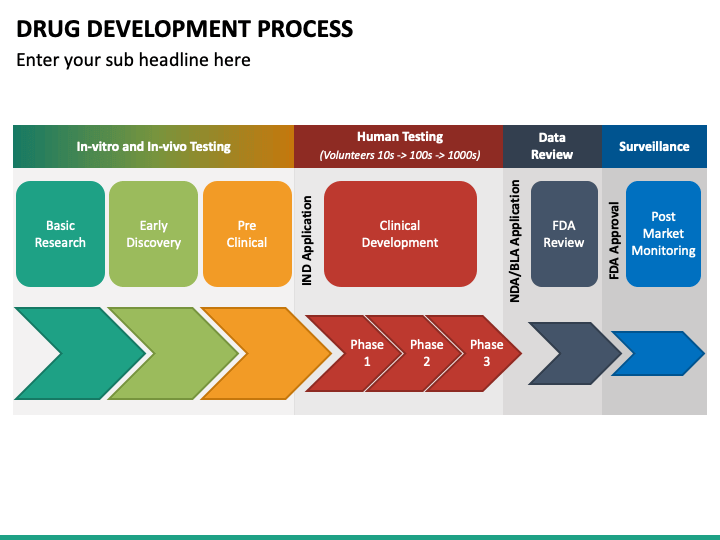
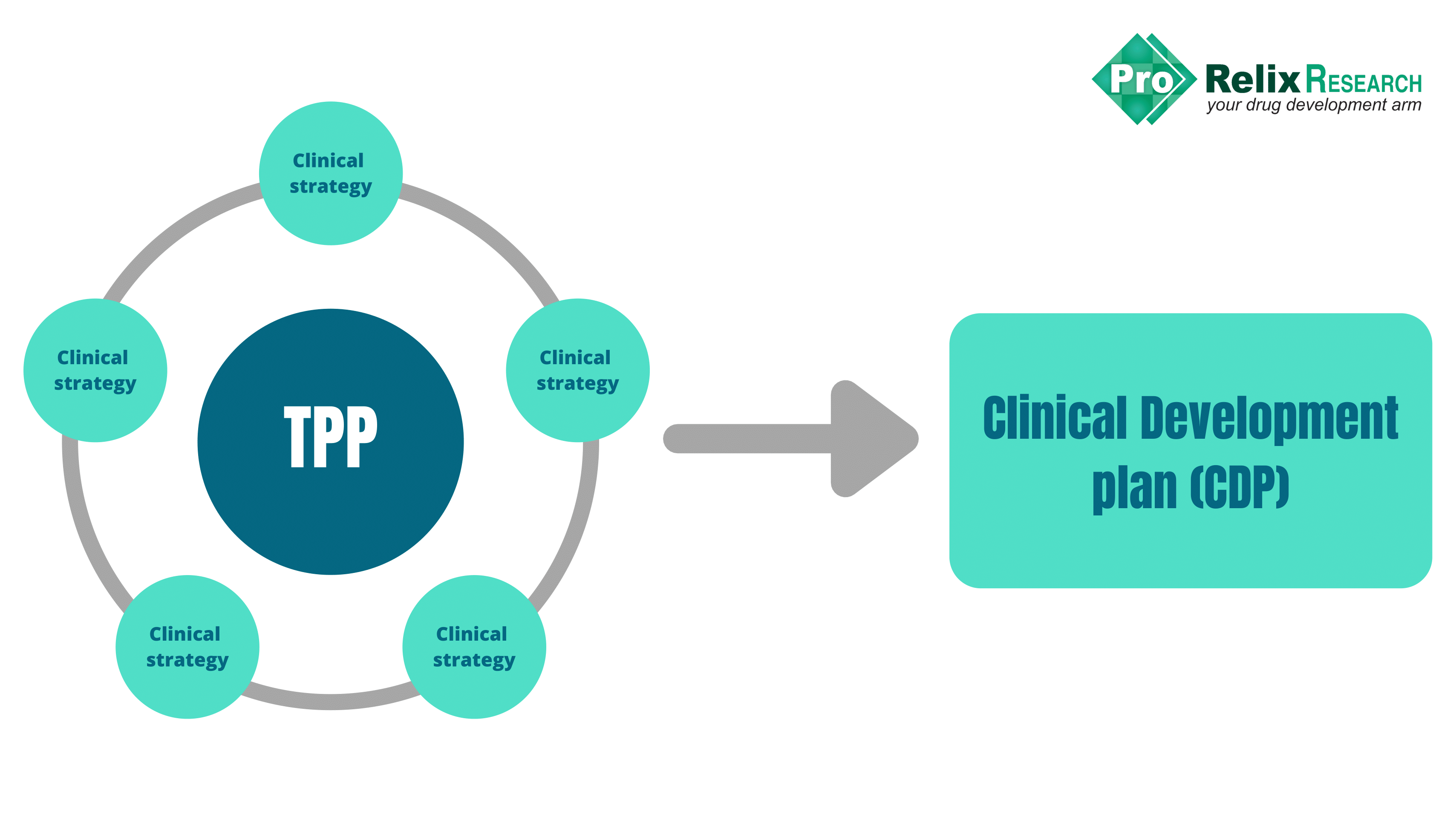
How to create a smart integrated drug development plan including considerations for cell and gene therapies - Biopharma Excellence

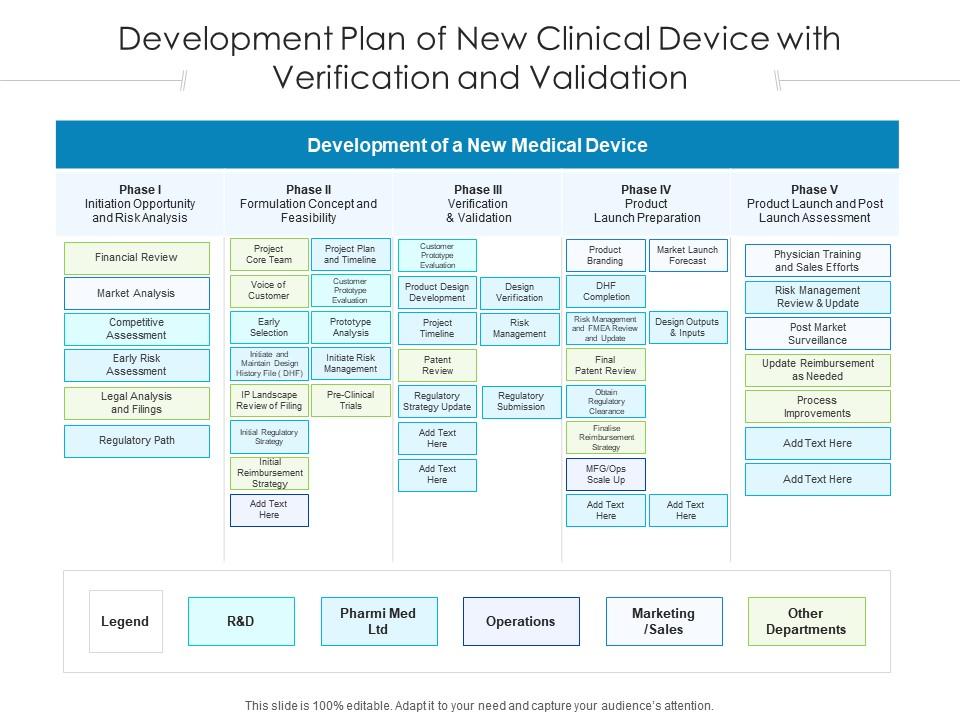
Clinical Drug Development Plan Lifecycle With Regulatory Approval | Presentation Graphics | Presentation PowerPoint Example | Slide Templates
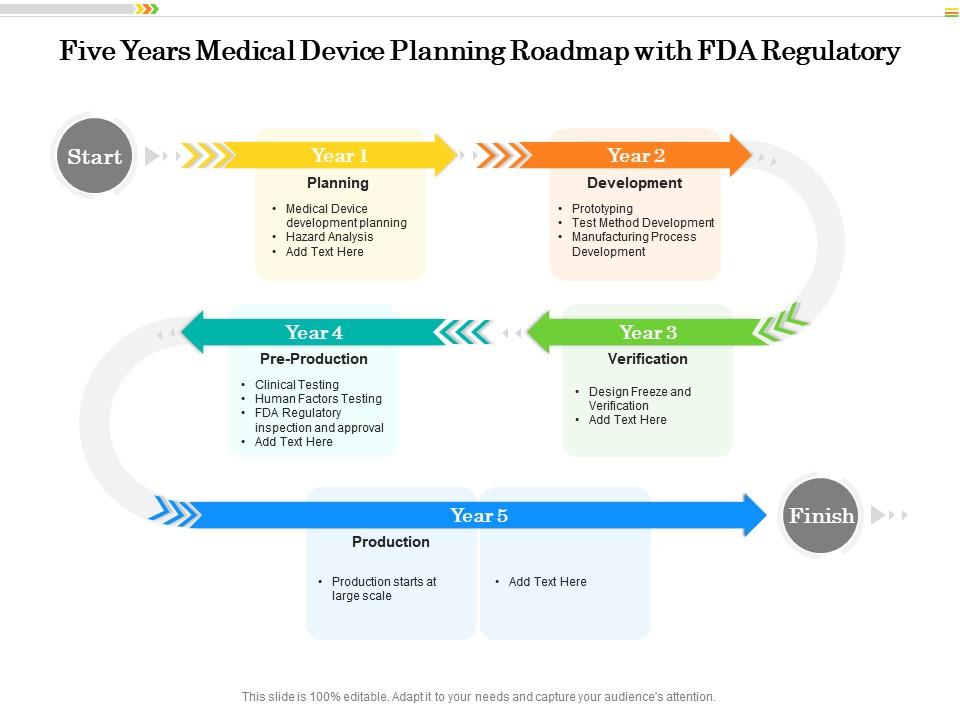
Five Years Medical Device Planning Roadmap With FDA Regulatory | Presentation Graphics | Presentation PowerPoint Example | Slide Templates